5th February 2013
GCSE Chemistry: Structures of Organic Compounds
Alkanes and Alkenes (Hydrocarbons)
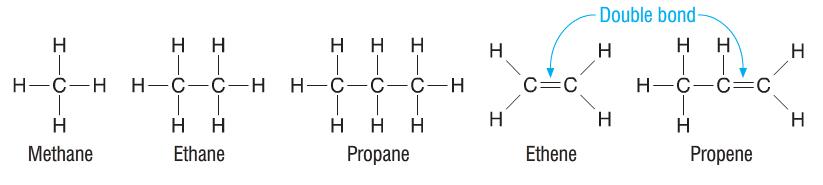
Organic compounds form the basis of all living things. Also, anything that contains a carbon (C) atom is considered an organic compound. Alkanes and alkenes (hydrocarbons) are simple examples of organic compounds. These two types of organic compounds are put together as follows:
Organic compounds form the basis of all living things. Also, anything that contains a carbon (C) atom is considered an organic compound. Alkanes and alkenes (hydrocarbons) are simple examples of organic compounds. These two types of organic compounds are put together as follows:
To work out how many carbon and hydrogen molecules there are in alkenes and alkanes, use the following formulas. For alkanes (saturated hydrocarbons) use: CnH2n+2. For alkenes (unsaturated hydrocarbons) use: CnH2n. For both formulas, n is the number of carbon atoms present. But what if you dont know how many carbon atoms there are in each type of hydrocarbon? I personally use a sequence of words. Monkeys Eat Peanut Butter. The first letter of each word is the first letter of each type of hydrocarbon. So Monkeys refers to Methane which has 1 carbon atom, Eat refers to Ethane which has 2 carbon atoms and so on. This method can also be used on more complex organic compounds such as alcohols as the stem remains constant. For organic compounds with over 4 carbon atoms, the pattern follows that of shapes. For example pentane, an alkane with 5 carbon atoms, is named after a pentagon which has 5 sides.
Alcohols
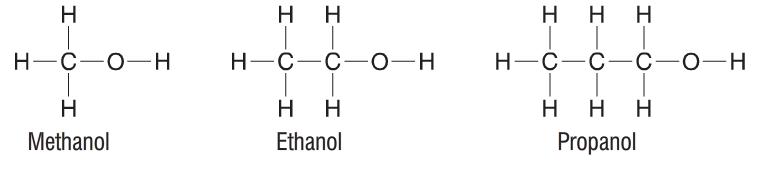
Alkanes and alkenes are simple in the fact that they contain only hydrogen and carbon atoms. Other, more complex, organic compounds contain other atoms such as oxygen (O) also. One very well known organic compound is alcohol. An alcohol is almost exactly the same as an alkane (saturated hydrocarbon), but it just contains one extra oxygen atom. This occurs when one of the hydrogen (H) atoms is replaced by an -OH group. A group such as the -OH group is often known as a functional group. The functional group (in this case the -OH group) gives each family of organic compounds its characteristics. Also, if a family of organic compounds have the same functional group, they are known as a homologous series. Alcohols are structured as follows:
Alcohols
Alkanes and alkenes are simple in the fact that they contain only hydrogen and carbon atoms. Other, more complex, organic compounds contain other atoms such as oxygen (O) also. One very well known organic compound is alcohol. An alcohol is almost exactly the same as an alkane (saturated hydrocarbon), but it just contains one extra oxygen atom. This occurs when one of the hydrogen (H) atoms is replaced by an -OH group. A group such as the -OH group is often known as a functional group. The functional group (in this case the -OH group) gives each family of organic compounds its characteristics. Also, if a family of organic compounds have the same functional group, they are known as a homologous series. Alcohols are structured as follows:
If you were ever asked to write down the formula of an alcohol when given a picture not unlike the ones above, many would simply just count up the carbon, hydrogen and oxygen atoms and write it like C2H6O (ethanol). Although this is technically correct, it is much better to use a structural formula. The structural formula of ethanol would be C2H5OH. By writing the formula like this you are able to see which funtional group is present in the compound and as such it is easier to identify what type of organic compound it is.
Carboxylic acids
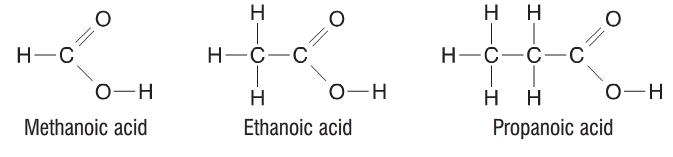
Although you might not think it, but there is another very well known organic compound in the form of a carboxylic acid. It's vinegar (which contains about 10% ethanoic acid)! Just like alcohols have the -OH functional group, carboxylic acids contain the -COOH functional group. The structure of carboxylic acids is as follows:
Carboxylic acids
Although you might not think it, but there is another very well known organic compound in the form of a carboxylic acid. It's vinegar (which contains about 10% ethanoic acid)! Just like alcohols have the -OH functional group, carboxylic acids contain the -COOH functional group. The structure of carboxylic acids is as follows:
Just like with alcohols, you wouldn't write the structural formula of ethanoic acid as C2H4O2, but instead as CH3COOH.
Esters
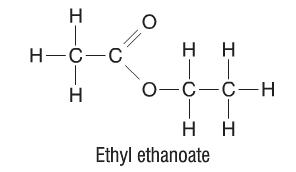
Esters are very similar to carboxylic acids. Esters are formed when we replace the H atom in the -COOH functional group with a hydrocarbon. As such, the functional group of esters becomes -COO-. The structure of an ester is as follows:
Esters
Esters are very similar to carboxylic acids. Esters are formed when we replace the H atom in the -COOH functional group with a hydrocarbon. As such, the functional group of esters becomes -COO-. The structure of an ester is as follows:
Once again, the structural formula of this ester would not be C4H8O2, but instead CH3COOC2H5 in order to highlight the functional group.
Share this:
HTML Comment Box is loading comments...